Virtual Data Room for M&A in 2026: 15-Document Checklist, Setup, and Pricing
Set up a virtual data room for M&A in 2026. Covers sell-side vs buy-side workflows, the 15-document checklist, folder structure, and top provider comparison.
A virtual data room for biotech is a secure online workspace used to manage the sensitive documentation that biotech and life sciences companies share during fundraising, licensing, M&A, clinical trials, and FDA regulatory filings. Biotech VDRs require stronger compliance than most industries: HIPAA, FDA 21 CFR Part 11, and GCP (Good Clinical Practice) all apply on top of the standard SOC 2, GDPR, and ISO 27001 baseline. Papermark supports biotech-grade data rooms at €99/month flat with a self-hostable deployment for regulated workloads.
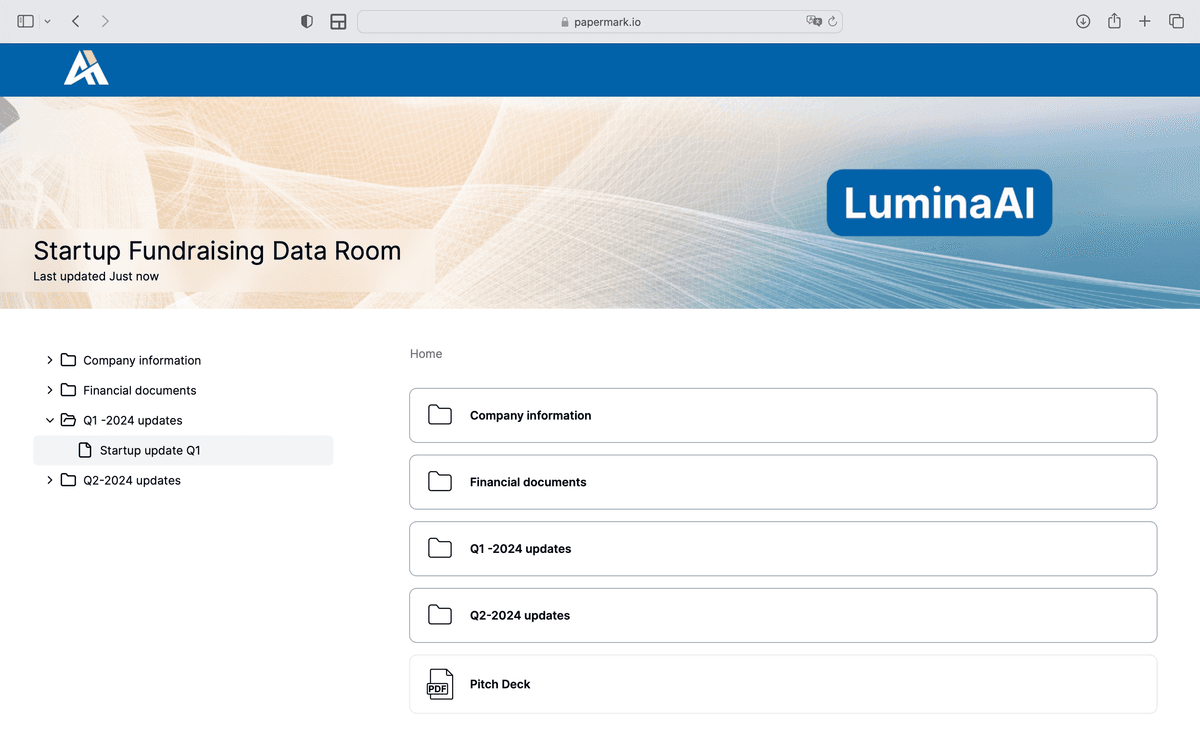
A biotech virtual data room is a secure online platform used by biotech, pharmaceutical, and life sciences companies to store, organize, and share confidential documents with investors, partners, regulators, and clinical trial sites. It provides granular permissions, dynamic watermarking, NDA enforcement, and the audit trail required for FDA 21 CFR Part 11 and HIPAA compliance.
Biotech differs from other industries in two important ways: documents routinely contain protected health information (PHI) or commercially priceless trade secrets (IP, clinical data, manufacturing processes), and the regulatory overlay is deeper than most sectors. A biotech VDR has to satisfy SOC 2 Type II and GDPR at minimum, with HIPAA and FDA 21 CFR Part 11 as common additions.
Biotech companies deal with documents where leakage has real legal, competitive, and regulatory consequences. Six specific reasons drive biotech firms to purpose-built VDRs.
Intellectual property protection. Patent applications, trade secrets, research data, and manufacturing processes represent the majority of a biotech company's value. Dynamic watermarking and granular permissions prevent leaks that would destroy a future patent filing.
Regulatory compliance. FDA 21 CFR Part 11 requires electronic records with audit trails, electronic signatures with identity verification, and system validation. HIPAA requires controlled access to PHI with BAA coverage for any vendor handling that data.
Licensing negotiations. Biotech licensing deals commonly involve 3-10 prospective partners, each with their own technical and legal teams. Scoped per-partner access plus NDA enforcement is essential.
M&A diligence. Biotech M&A involves massive technical document sets (clinical trial data, patent portfolios, manufacturing records) shared with strategic and PE buyers.
Clinical trial management. Multi-site trials require access to protocols, informed consent forms, site agreements, and safety data across investigators, monitors, and sponsors.
Investor fundraising. Biotech Series A/B rounds involve detailed scientific and clinical diligence, often with investors who bring their own scientific advisors into the data room.
Biotech data rooms add 7-10 categories to the standard fundraising or M&A document set. The table below lists all essential biotech documents.
| Document | Category | Essential | Nice to Have |
|---|---|---|---|
| Confidential Information Memorandum (CIM) | Company Overview | ✔️ | |
| Executive Summary | Company Overview | ✔️ | |
| Scientific Advisory Board Information | Company Overview | ✔️ | |
| Financial Statements | Financial Information | ✔️ | |
| Tax Records | Financial Information | ✔️ | |
| Incorporation Documents | Legal Documents | ✔️ | |
| Shareholder Agreements | Legal Documents | ✔️ | |
| Patent Applications and Approvals | Intellectual Property | ✔️ | |
| IP Licensing Agreements (in and out) | Intellectual Property | ✔️ | |
| Trade Secret Policies | Intellectual Property | ✔️ | |
| IND / NDA / BLA Filings | Regulatory | ✔️ | |
| FDA Correspondence | Regulatory | ✔️ | |
| Regulatory Approvals (per jurisdiction) | Regulatory | ✔️ | |
| Clinical Trial Protocols | Clinical Research | ✔️ | |
| Clinical Trial Data and Results | Clinical Research | ✔️ | |
| Informed Consent Forms | Clinical Research | ✔️ | |
| Investigator Brochures | Clinical Research | ✔️ | |
| Safety Data / Pharmacovigilance | Clinical Research | ✔️ | |
| Manufacturing Process Documentation | Manufacturing | ✔️ | |
| Quality Control Procedures (cGMP) | Manufacturing | ✔️ | |
| Pipeline Projects and Roadmap | Research and Development | ✔️ | |
| Research and Development Reports | Research and Development | ✔️ | |
| Customer and Supplier Contracts | Operational | ✔️ | |
| Employee Agreements and Key-Person | Human Resources | ✔️ | |
| Partnerships and Collaborations | Operational | ✔️ | |
| Market Analysis | Market and Competitive | ✔️ | |
| Competitive Analysis | Market and Competitive | ✔️ | |
| Risk Factors | Additional | ✔️ | |
| Use of Funds | Additional | ✔️ |
Biotech VDRs operate under a deeper compliance stack than most industries. Four frameworks routinely apply.
HIPAA (Health Insurance Portability and Accountability Act). Required any time the data room contains protected health information (PHI). Requires a Business Associate Agreement (BAA) with the VDR vendor, audit logging, encryption at rest and in transit, and access controls. Papermark supports HIPAA-ready deployments via self-hosting plus signed BAA on enterprise plans.
FDA 21 CFR Part 11. US FDA regulation covering electronic records and electronic signatures. Requires audit trails showing who created, modified, or viewed records; electronic signatures with identity verification; system validation documentation; and controls to prevent unauthorized record alteration. Applicable to IND, NDA, BLA, and related filings.
GCP (Good Clinical Practice). ICH E6(R2) and related guidelines covering clinical trial conduct, documentation, and monitoring. The VDR supports GCP workflows by maintaining immutable audit logs, version histories, and scoped access for investigators, monitors, and sponsors.
GDPR and regional data residency. EU-conducted trials require GDPR alignment and often EU data residency. Multi-jurisdiction trials need per-site access controls that respect local privacy law.
YYYY-MM-DD_DocumentType_Indication.Essential biotech security controls:
Different stages of a biotech company's lifecycle require different data room configurations. The table below maps the five most common deal stages to the documents and access patterns they call for.
| Deal stage | Primary audience | Key document categories | Access pattern |
|---|---|---|---|
| Seed / Pre-IND | VC investors, family offices | Pipeline R&D, scientific founders, IP filings | View-only with watermark, NDA gated |
| Series A/B funding | Institutional VCs, life sciences funds | Financial model, IND filing, clinical strategy | Per-investor scoped folders, Q&A module |
| Pre-clinical to IND | FDA, IRB, contract research orgs | IND application, IB, study protocols, GLP documents | Compliance-grade audit trail (21 CFR Part 11) |
| Clinical trial (Phase I-III) | CROs, sponsors, IRBs, monitors | Study protocols, ICFs, CRFs, monitoring reports, SAE reports | GCP-compliant per-site access, electronic signatures |
| Licensing / partnership | Big pharma BD, licensing partners | IND/NDA, manufacturing, IP, commercial strategy | Multi-bidder isolation, dynamic watermarking |
| M&A / acquisition | Strategic acquirers, PE | Full corporate diligence + biotech-specific (regulatory, clinical) | Per-bidder scoped, structured Q&A |
| IPO | SEC, audit firms, underwriters | S-1 disclosures, audited financials, regulatory pipeline | Full audit trail, multi-team review |
For a workflow-specific guide, see data room for IPO and virtual data room for M&A.
Each regulation maps to specific technical and procedural controls. The table below makes the mapping explicit.
| Regulation | Requirement | VDR feature |
|---|---|---|
| HIPAA Security Rule | Access controls | Role-based access, MFA, scoped folders |
| HIPAA Security Rule | Audit controls | Append-only audit log, immutable retention |
| HIPAA Security Rule | Integrity | Document version control, hash verification |
| HIPAA Security Rule | Transmission security | TLS 1.3, AES-256 at rest |
| HIPAA Privacy Rule | Minimum necessary | Per-recipient scoped permissions |
| HIPAA / BAA | Business Associate Agreement | Signed BAA on enterprise plans |
| 21 CFR Part 11 | Electronic signatures | Identity-verified signature with timestamp |
| 21 CFR Part 11 | Audit trail | Append-only log of all create/modify/view events |
| 21 CFR Part 11 | System validation | Documented IQ/OQ/PQ on self-hosted deployments |
| 21 CFR Part 11 | Record protection | Read-only document storage, version history |
| GCP / ICH E6(R2) | Document version control | Version history with timestamps |
| GCP / ICH E6(R2) | Per-site / per-investigator access | Scoped folder permissions |
| GCP / ICH E6(R2) | Monitor and sponsor access | Audit-log-supported review workflow |
| GDPR (EU trials) | Data residency | EU/Frankfurt hosting, self-hosted EU option |
| GDPR (EU trials) | Lawful basis documentation | DPA on file, retention policies |
For platform-level details, see Papermark GDPR compliance and Papermark SOC 2 compliance.
Five mistakes show up repeatedly in biotech diligence and licensing.
1. Mixing PHI with non-PHI documents. Once any document in the room contains protected health information, the entire room must meet HIPAA controls. Either segregate PHI into a HIPAA-scoped sub-room or apply HIPAA controls to the full room.
2. Skipping the BAA before sharing PHI. The Business Associate Agreement must be signed before PHI lands in the data room, not retroactively. Verify with the platform vendor.
3. Letting clinical site investigators see other sites' data. GCP requires per-site scoping. A blanket "all investigators" permission group is non-compliant. Configure per-site folders.
4. Treating IND/NDA as static documents. IND and NDA filings evolve through FDA correspondence cycles. Use document version control and date-stamped folders, not folder rewrites.
5. Skipping the validation documentation on self-hosted deployments. 21 CFR Part 11 requires Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ) for the system handling regulated records. Document these before going live.
Out-licensing pipeline assets to big pharma. Biotech companies with one or two clinical-stage assets often partner with pharma for late-stage development and commercialization. The data room hosts the IND, IB, clinical trial results, manufacturing cGMP documentation, IP portfolio, and commercial strategy. Multiple pharma BD teams may evaluate in parallel - per-bidder scoped permissions and dynamic watermarking are mandatory.
M&A of clinical-stage biotechs. Strategic acquirers typically run 4-8 weeks of diligence on full corporate documentation plus biotech-specific (regulatory pipeline, clinical data, manufacturing). The data room must support 1,500-3,000 documents and 30-50 reviewers under deal-grade pressure.
Series B/C and crossover investor diligence. Life sciences funds (RA Capital, ArrowMark, Perceptive, Cormorant) run institutional diligence including pipeline review, financial model validation, IP freedom-to-operate, and management reference checks. Q&A module with logged answers preserves the audit trail for follow-on rounds.
Investigator-initiated trials. Academic medical centers and biotechs co-running investigator-initiated trials need per-site scoped access to clinical protocols, ICFs, and adverse event reports. GCP-compliant audit trails preserve regulatory accountability.
Combination product (drug + device) submissions. FDA submissions for combination products require both 21 CFR Part 11 (drug record integrity) and FDA medical device QSR documentation. The data room must support both sets of audit-log requirements.
Biotech-specific pricing tends to be higher than general VDR pricing because of the compliance overhead. Typical 2026 ranges:
| Provider | Entry tier (biotech-ready) | Notes |
|---|---|---|
| Papermark | €99/month flat (HIPAA via self-hosted + BAA enterprise) | Self-hostable for clinical site or sponsor on-prem requirements |
| Firmex | $625/month flat | Industry-standard for biotech licensing rooms |
| iDeals | Custom (~$10,000+/year) | Per-page or custom annual contracts |
| Datasite | Custom ($25,000+/year) | Common for late-stage biotech M&A |
| Intralinks | Custom ($25,000+/year) | Common for IPO-track biotechs |
For the full pricing breakdown, see virtual data room cost in 2026.
Papermark supports biotech deployments at €99/month flat for the Data Rooms plan, with a self-hostable open-source version for regulated workloads requiring on-prem hosting: